1.0 OBJECTIVE
To provide the procedure for Estimation of Famotidine from Tablet Formulations
2.0 SCOPE
This procedure is Applicable to the Estimation of Famotidine from Tablet Formulations
3.0 RESPONSIBILITY
3.1 QC-Chemist
4.0 ACCOUNTABILITY
4.1 Executive – Quality control
4.2 Head – Quality control
5.0 PROCEDURE
5.1Label Claim:
Famotidine Tablets IP/USP ...... 40mg
5.2 Assay Method:
5.2.1 Standard solution:
Accurately weigh about 20mg of Famotidine working standard and transfer it into 100ml of volumetric flask, add 50 ml of double distilled water, shake well until the material gets dissolved and make up to 100ml with above solution.
5.2.2 Sample solution:
Weigh and powder 20 tablets. Weigh accurately the tablets powder equivalent to about 20mg of Montelukast sodium into 100ml of volumetric flask, add 50ml of double distilled water, shake well until dissolve the substance or Sonicate for 3 minutes, make up to volume with double distilled water.
Filter the sample Solution with whatman filter paper and reject first few ml of the filtrate.
Measure the absorbance at about 260nm using double distilled water as blank
5.2.3 Calculate the content by comparison.
= ____________x100
LC
LC: Label Claim
6.0 ANNEXURES
Nil




.JPG)













![Difference between Stability[Shelf life] Specification and Release Specification](https://4.bp.blogspot.com/-O3EpVMWcoKw/WxY6-6I4--I/AAAAAAAAB2s/KzC0FqUQtkMdw7VzT6oOR_8vbZO6EJc-ACK4BGAYYCw/w680/nth.png)
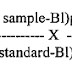
0 Comments