1.0 OBJECTIVE
To
lay down the procedure for the
2.0 SCOPE
This
SOP is applicable to
3.0 BACKGROUND
NIL
4.0 RESPONSIBILITY
4.1
QC-Chemist
4.2
Head-QC
5.0 PROCEDURE
5.1 For
this i have taken residual solvents(Ethanol, IPA, Acetonitrile, Ethyl
acetate, Toluene, Acetic acid) in the memantine Hydrochloride.
5.2 Chromatographic Conditions:
Instrument
is Agilent 6890 N, Column AT-624, 30x 0.53x 3 microns, injector
Temperature is 140 degrees, split is 1:5, Detector Temerature 260
degrees(FID), Carrier gas is helium, Column flow is 2.8 PSI, Load is 1
microleter, diluent is N,N dimethyl acetamide and Column is programmed in the following way
| Rate(degree/min) | Temperature(degrees) | Hold Time(Min) |
|---|---|---|
| -- | 40 | 10 |
| 6 | 130 | 5 |
| 35 | 240 | 11 |
5.4 Standard Preparation:
Solution -I:
Transfer 127 microliters of Ethanol, 127 microliters of IPA, 11 microliters of Acetonitrile, 111 microliters of Ethyl acetate, 21 microliters of Toluene, 95 microliters of Acetic acid in 10 ml
volumetric flask dilute with diluent.
Solution-II:
transfer 0.1 ml of solution-I into 10 ml Volumetric flask. This
solution gives 5000 ppm of ethanol, 5000 PPM of IPA, 410 PPM of ACN,
5000 PPM of ethyl Acetate, 890 PPM of Toluene, and 5000 PPM of acetic
acid with respect to 20mg/ml of sample preparation.
5.5 Calculate each solvent with following the formula
B1 is area of solvent in the blank
5.6 Typical chromatograms for Standard and Sample solution:
 |
| Standard Solution Chromatogram |
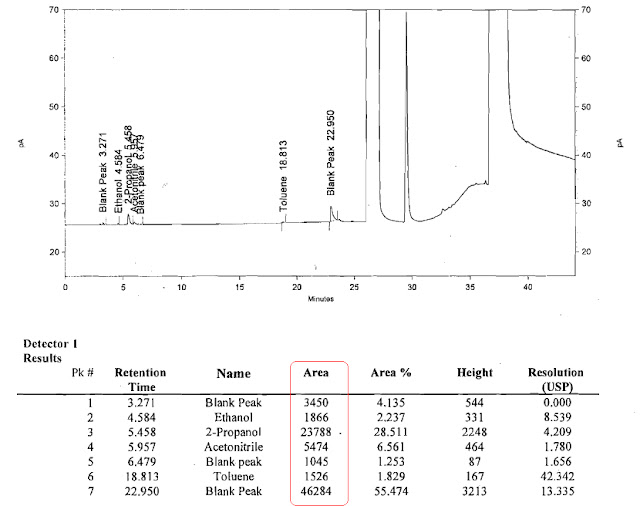 |
| Sample solution Chromatograms |
5.7 Let us start the calculation
Standard Areas of 2 injections(After subtracting from Blank)
| STD | Ethanol | IPA | Acetonitrile | Ethyl acetate | Toluene | Acetic acid |
| 169889 | 186930 | 22551 | 149725 | 75567 | 77700 | |
| 170012 | 185890 | 22689 | 146678 | 73212 | 76890 | |
| sum | 339901 | 372820 | 45240 | 296403 | 148779 | 154590 |
| Average | 169950.5 | 186410 | 22620 | 148201.5 | 74389.5 | 77295 |
Samples area of two injections (After subtracting from Blank)
| Sample | Ethanol | IPA | Acetonitrile | Ethyl acetate | Toluene | Acetic acid |
| 1866 | 23788 | 5733 | 0 | 1526 | 0 | |
| 1922 | 22674 | 5660 | 0 | 1634 | 0 | |
| sum | 3788 | 46462 | 11393 | 0 | 3160 | 0 |
| Average | 1894 | 23231 | 5696.5 | 0 | 1580 | 0 |
5.8 Solvent densities:
Weight of the sample: 199.98
dilution of the sample : 20 mg /ml
Ethanol =
Ethanol =
(1894/ 169950) * (127 micro liters x0.789 g/ml /10) * (0.1/10) * (20 mg/ml/ 199.98 mg) * 1000000
= 56.48 PPM
IPA=
(23231/ 186410) * (127 micro liters x0.785 g/ml /10) * (0.1/10) * (20 mg/ml/ 199.98 mg) * 1000000
= 621.27 PPM
Like that you can calculate PPM obtained for other solvents
6.0 ANNEXURES
Nil









.JPG)



![Cleaning Validation Calculations-Maximum Allowable Carryover[MACO]](https://blogger.googleusercontent.com/img/b/R29vZ2xl/AVvXsEiGzQe3wNgJT58HARMOttesykz3va8OngShcTqWy5cOaalJxRSsMcfFzhe1UxZXWTdrCPrcXy5za2onmmI8DSJ2vgkxE7mZQot1dZkAnQ11KCcqCL59rZCtIgWyAKFCo82hiuYZyJ8N6tKIx5WeVkY8nqoB3XVTGLGYb0lSZ4VfQtd8vBJDPWLRYvz2fA/w72-h72-p-k-no-nu/HBEL.jpg)





0 Comments