1.0 OBJECTIVE
To provide the procedure
for the determination mass balance ratio in stress studies
2.0 SCOPE
This procedure is
Applicable to the determination of mass balance ratio in stress studies
3.0 RESPONSIBILITY
3.1 QC-Chemist
4.0 ACCOUNTABILITY
4.1 Executive – Quality
control
4.2 Head – Quality
control
5.0 PROCEDURE
5.1 Mass balance correlates
the measured loss of a parent drug to the measured increase in the amount of
degradation products. It is a good quality control check on analytical methods
to show that all degradation products are adequately detected and do not interfere
with quantitation of the parent drug (i.e., stability-indicating methods).
5.2 How much
Degradation?
Usually, 5 % to 10 % of
degradation is looked for as a forced degradation study during the different
forced degradative conditions. There is not all compounds/formulations will
degrade by 10% under even extreme conditions, however, a sufficient attempt must
be taken to promote degradation. Any stability observed from the studies must
be scientifically explained and justified in case of low degradation levels
(i.e. < 10%). These types of criteria can be used within internal guidance
documents where extreme conditions are defined.
There is no guideline or
industry-accepted practice for mass balance limit for forced degradation
studies but we need to demonstrate the mass balance of the method, whatever is
achieved if all other validation parameters are OK. Generally, 95 % to 105 %
mass balance is well accepted.
Total mass
= (Amount of mass remained + Amount of known degradants + Amount of unknown
degradants)
So, here I will convert the equation for better understanding. After each degradation study,
5.5 Factors effecting the Mass balance Ratio
5.5.1Relative response factors play an important role to calculate the exact mass balance.
5.5.2 The molecular weight
of Drugs and the molecular weight of the degradant formed plays an important
role to calculate the exact mass balance.
5.5.3 Poor recovery of Parent or Drug product
5.5.4 Chromatographic issues in drug and degradants
![]()
6.0
ANNEXURES
Nil




.JPG)













![Difference between Stability[Shelf life] Specification and Release Specification](https://4.bp.blogspot.com/-O3EpVMWcoKw/WxY6-6I4--I/AAAAAAAAB2s/KzC0FqUQtkMdw7VzT6oOR_8vbZO6EJc-ACK4BGAYYCw/w680/nth.png)
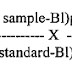
0 Comments