1.0 OBJECTIVE
To provide the procedure for Estimation of Diclofenac Sodium tablets (50mg) formulation.
2.0 SCOPE
This procedure is Applicable to the Estimation of Diclofenac Sodium tablets (50mg) formulation.
3.0 RESPONSIBILITY
3.1 QC-Chemist
4.0 ACCOUNTABILITY
4.1 Executive – Quality control
4.2 Head – Quality control
5.0 PROCEDURE
5.1Label Claim:
Each film coated tablet contains
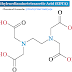
Diclofenac Sodium IP/BP-------- 50mg
5.2 Assay Method:
5.2.1 Preparation of Buffer Solution
Acetate buffer of pH 5.5 was prepared by weighed 5.48 gm of sodium acetate dissolved in 900 ml distilled water in 1000 ml volumetric flask. Add 10 ml of 2N glacial acetic acid (0.6 ml glacial acetic acid in 10 ml distilled water). The volume was made up with distilled water to get a final solution of pH 5.5.
5.2.1 Standard solution:
The standard stock solution of diclofenac sodium was prepared by transferring, accurately weighed 100 mg of drug powder were transfer to 100 ml volumetric flasks and dissolved in acetate buffer pH 5.5 (50 ml). The flask were sonicated for 15 min at 40°C to give clear solution and volume were made up to mark with Acetate buffer to give a solution. From this 10 ml of the solution was transferred to a 100 ml volumetric flask and make up the volume with same solvent.
5.2.2 Sample solution:
Weigh and powder 20 tablets. Weigh accurately the tablets powder equivalent to about 50mg of Amlodipine besylate into 50ml of volumetric flask, add 10ml of acetate buffer pH 5.5 , shake well until dissolve the substance or Sonicate for 3 minutes, make up to volume with acetate buffer pH 5.5 .
Filter the sample Solution with whatman filter paper and reject first few ml of the filtrate.
From this 10 ml of the solution was transferred to a 100 ml volumetric flask and make up the volume with same solvent.
Measure the absorbance at about 276nm using double distilled water as blank
5.2.3 Calculate the content by comparison.
= ____________x100
LC
LC: Label Claim
6.0 ANNEXURES
Nil





.JPG)











![Difference between Stability[Shelf life] Specification and Release Specification](https://4.bp.blogspot.com/-O3EpVMWcoKw/WxY6-6I4--I/AAAAAAAAB2s/KzC0FqUQtkMdw7VzT6oOR_8vbZO6EJc-ACK4BGAYYCw/w680/nth.png)
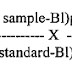
0 Comments